Simultaneous Determination of Benzoic Acid, Sorbic Acid, Sodium Saccharin, and Acesulfame-K in Food
- Analyte: Benzoic acid, Sorbic acid, Sodium saccharin, Acesulfame-K
- System: EClassical 3200 HPLC System
- Column: Supersil AQ-C18 (5μm, 4.6×250mm)
- Highlight: Simultaneous detection of four food additives with high sensitivity.
Introduction
Food additives play a crucial role in preserving food quality, safety, and flavor. Among them, preservatives like benzoic acid and sorbic acid, and sweeteners like sodium saccharin and acesulfame-K are widely used. To ensure their "reasonable" and "moderate" use, reliable detection methods are essential. Elite company presents an HPLC-based solution for the simultaneous determination of these four additives. This method not only allows concurrent analysis but also addresses the issue of short lifetime commonly encountered with conventional C18 columns.
Standards and Reagents
Standards: benzoic acid (1000 μg/mL), sorbic acid (1000 μg/mL), sodium saccharin (1000 μg/mL), acesulfame-K (>97.0%)
Reagents: methanol (HPLC grade), ammonium acetate (analytical grade), deionized water (18.2 MΩ·cm)
Other materials: potassium ferrocyanide solution, zinc acetate solution, n-hexane (for high-fat samples), ammonia solution (1+99), ethanol.
Standard Solution Preparation
Mixed Standard Stock Solution (50 μg/mL): Accurately weigh appropriate amounts of benzoic acid, sorbic acid, sodium saccharin, and acesulfame-K into a volumetric flask. Dissolve and dilute to volume with deionized water to achieve a final concentration of 50 μg/mL for each. Mix well, filter through a 0.45 μm membrane, and store at 4°C.
Working Solutions: Dilute the stock solution with deionized water to desired concentrations as needed.
Sample Pretreatment
Three types of sample pretreatment are described based on sample matrix:
General Samples (e.g., beverages, fruit juices):
Accurately weigh about 2 g (to 0.001 g) of sample into a 50 mL centrifuge tube. Add ~25 mL water, vortex mix, and sonicate in a 50°C water bath for 20 min. Cool to room temperature, add 2 mL potassium ferrocyanide solution and 2 mL zinc acetate solution, mix, and centrifuge at 8000 r/min for 5 min. Transfer the aqueous phase to a 50 mL volumetric flask. Add 20 mL water to the residue, vortex, sonicate for 5 min, centrifuge again, and combine the supernatants. Dilute to volume with water, mix, and filter through a 0.22 μm membrane before HPLC analysis. (Note: For carbonated beverages, fruit wines, and distilled spirits, the protein precipitation step may be omitted.)
Samples Containing Gums (e.g., jelly, candy):
Weigh about 2 g sample into a centrifuge tube, add ~25 mL water, vortex, and heat in a 70°C water bath to dissolve. Then sonicate at 50°C for 20 min. Proceed with protein precipitation and centrifugation as described for general samples.
High-Fat Samples (e.g., chocolate, cream, fried foods):
Weigh about 2 g sample into a centrifuge tube, add 10 mL n-hexane, and heat in a 60°C water bath for ~5 min with occasional shaking to dissolve fat. Add 25 mL ammonia solution (1+99) and 1 mL ethanol, vortex, and sonicate at 50°C for 20 min. Cool, add protein precipitants, centrifuge, discard the organic phase, and transfer the aqueous phase to a 50 mL volumetric flask. Re-extract the residue once more and combine the aqueous phases. Dilute to volume and filter.
Instruments and Equipment
HPLC system: EClassical 3200 configured with D3210 UV-Vis detector, P3220 high-pressure pump, O3220 column oven, S3210 autosampler, T3200 solvent bottle tray, Kromstation CDS.
Supersil AQ-C18 column (5 μm, 4.6×250 mm)
Pretreatment equipment: Solvent filter assembly, diaphragm vacuum pump, ultrasonic cleaner, precision electronic balance (0.1 mg sensitivity), centrifuge, volumetric flasks, micropipettes, etc.
Chromatographic Conditions
Column: Supersil AQ-C18 (5 μm, 4.6×250 mm)
Mobile Phase: Methanol / 0.02 mol/L ammonium acetate solution = 5:95 (V/V)
Flow Rate: 1.0 mL/min
Detection: UV at 230 nm
Injection Volume: 10 μL
Column Temp.: 30°C
Experimental Results
Standard Separation
A typical chromatogram of a mixed standard solution at 10.0 μg/mL is shown in Figure 1. The chromatographic parameters are summarized in Table 1.
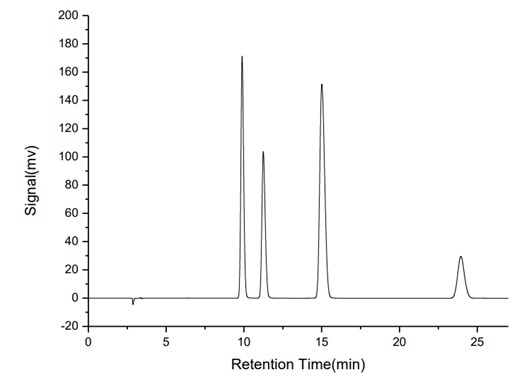
Fig.1 Chromatogram of mixed standard solution (5μg/mL) of benzoic acid, sorbic acid, sodium saccharin, and acesulfame-K
Table 1. Chromatographic parameters
| Compound |
Retention Time (min) |
Peak Area (mV·sec) |
Asymmetry |
Plate Number |
| Acesulfame-K |
9.88 |
2143.67 |
1.30 |
14231 |
| Benzoic Acid |
11.25 |
1551.17 |
1.42 |
12961 |
| Sorbic Acid |
15.01 |
3402.26 |
1.46 |
9808 |
| Sodium Saccharin |
23.95 |
914.15 |
1.16 |
13789 |
Linearity and Detection Limit
Calibration curves were constructed using standard working solutions at concentrations of 5, 10, 20, 30, 40, 50, and 100 μg/mL. Linear regression equations and correlation coefficients are listed in Table 2. The instrument detection limits (S/N=3) and quantification limits (S/N=10) are given in Table 3.
Table 2. Linear equations and correlation coefficients
| Compound |
Linear Equation |
Correlation Coefficient (R) |
| Acesulfame-K |
y = 211.62x + 29.85 |
0.9999 |
| Benzoic Acid |
y = 152.94x + 22.34 |
0.9999 |
| Sorbic Acid |
y = 335.49x + 49.76 |
0.9999 |
| Sodium Saccharin |
y = 90.11x + 12.89 |
0.9999 |
Table 3. Detection and quantification limits
| Compound |
Detection Limit (μg/mL) |
Quantification Limit (μg/mL) |
| Acesulfame-K |
0.0054 |
0.0180 |
| Benzoic Acid |
0.0086 |
0.0287 |
| Sorbic Acid |
0.0053 |
0.0177 |
| Sodium Saccharin |
0.0314 |
0.1047 |
The method exhibits excellent linearity and low detection limits, making it suitable for routine monitoring of these additives in various food matrices.

