Tylosin tartrate is a macrolide antibiotic widely used in veterinary medicine for the treatment of respiratory and gastrointestinal infections. It is also employed as a growth promoter in livestock. Accurate determination of tylosin tartrate is essential for quality control of pharmaceutical formulations. This report presents an HPLC method for the analysis of tylosin tartrate using a SinoChrom ODS-BP column with gradient elution and UV detection at 254 nm.
Standards: Tylosin tartrate (CAS 1405-54-5)
Reagents: deionized water (18.2 MΩ·cm), methanol (HPLC grade), acetonitrile (HPLC grade), ammonium acetate (analytical grade), acetic acid (glacial, analytical grade). Other solvents and chemicals as required for sample pretreatment (e.g., extraction solvents, filtration membranes)
Standard Solution Preparation
Accurately weigh about 10 mg of tylosin tartrate standard into a 10 mL volumetric flask. Dissolve in methanol, dilute to volume with the same solvent, and mix well. Filter through a 0.45 μm membrane before injection.
For test samples, follow the same procedure as for the standard solution: accurately weigh an appropriate amount of sample (equivalent to about 10 mg of tylosin tartrate), dissolve in methanol, dilute to 10 mL, and filter through a 0.45 μm membrane.
HPLC System: EClassical 3200 equipped with quaternary pump, UV-vis detector, autosampler, column oven, solvent bottle tray, Kromstation CDS.
Pretreatment equipment: analytical balance (0.0001 g sensitivity), ultrasonic cleaner, centrifuge, solvent filtration apparatus, membrane filters, pipettes, etc.
Column: SinoChrom ODS-BP (5μm, 4.6×250 mm)
Mobile Phase: A: 0.01 mol/L ammonium acetate solution (pH 3.0 adjusted with acetic acid); B: acetonitrile, in gradient (Table 1)
Flow Rate: 1.0 mL/min
Detection: UV at 254 nm
Injection Vol.: 20 μL
Column Temp.: 30°C
Table 1. Gradient program
A representative chromatogram of tylosin tartrate under the described conditions is shown in Figure 1 (refer to original document). The peak shape is satisfactory, and the retention time is reproducible. The gradient program ensures adequate separation of tylosin tartrate from any related substances.
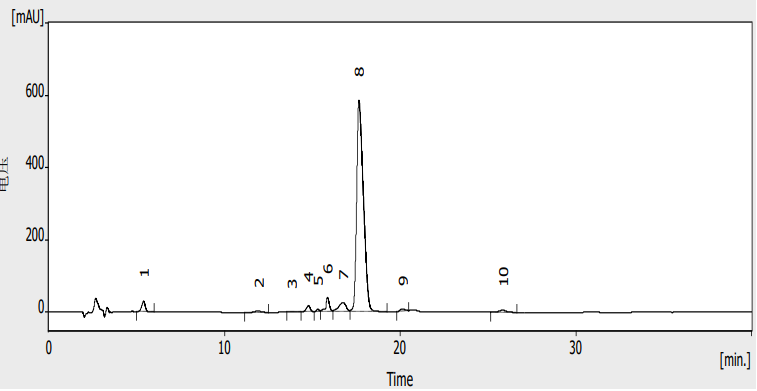
Fig.1. Ceftiofur peak parameters – SinoPak BEH T-C18 column (1st vs. 13th injection)

