HPLC Determination of Rutin in Mulberry Leaves
- Analyte: Rutin
- System: EClassical 3200 HPLC System with Diode Array Detector (DAD)
- Column: Hypersil ODS2 (5μm, 4.6×150 mm)
- Highlight: A reliable HPLC method for rutin analysis in mulberry leaves with good peak shape, meeting pharmacopoeial requirements.
Introduction
Rutin, also known as rutoside or vitamin P, is a natural flavonol glycoside widely present in plants. It exhibits various pharmacological activities including anti-inflammatory, antioxidant, anti-allergic, and antiviral effects. In traditional Chinese medicine, mulberry leaves are a common source of rutin. According to pharmacopoeial methods, proper chromatographic separation and peak shape are essential for accurate quantification. This report presents an HPLC method for the determination of rutin in mulberry leaves using an EClassical 3200 system, addressing issues such as solvent effects and column selection to achieve satisfactory peak performance.
Standards and Reagents
Standards: Rutin (CAS 153-18-4)
Reagents: deionized water (18.2 MΩ·cm), methanol (HPLC grade), phosphoric acid (AR), other solvents and chemicals as required for sample pretreatment (e.g., extraction solvents, filtration membranes)
Standard Solution Preparation
Provided by the client (solvent, methanol). To avoid solvent effects, the standard was diluted 5-fold with the initial mobile phase composition (methanol/0.5% phosphoric acid = 30/70) before injection.
All solutions are filtered through a 0.45 μm membrane before injection
Sample Pretreatment
Provided by the client (solvent, methanol).
Instruments and Equipment
HPLC System: EClassical 3200 equipped with quaternary pump, DAD, autosampler, column oven, solvent bottle tray, Kromstation CDS.
Pretreatment equipment: analytical balance (0.0001 g sensitivity), ultrasonic cleaner, centrifuge, solvent filtration apparatus, membrane filters, pipettes, etc.
Chromatographic Conditions
Column: Hypersil ODS2 (5μm, 4.6 × 150 mm)
Mobile Phase: A: Methanol, B: 0.5% phosphoric acid aqueous solution, in gradient (Table 1).
Flow Rate: 1.0 mL/min
Detection: UV at 358 nm
Injection Vol.: 10 μL
Column Temp.: 30°C
Table 1. Gradient program
| Time (min) |
A% |
B% |
| 0 |
30 |
70 |
| 5 |
30 |
70 |
| 10 |
35 |
65 |
| 15 |
40 |
60 |
| 18 |
50 |
50 |
| 18.1 |
30 |
70 |
| 28 |
30 |
70 |
Results and Discussion
Standard Separation
The rutin standard solution (diluted 5-fold with initial mobile phase) was analyzed under the above conditions. A representative chromatogram is shown in Figure 1. The retention time was approximately 14.356 min, with good peak shape. Chromatographic parameters are summarized in Table 2.
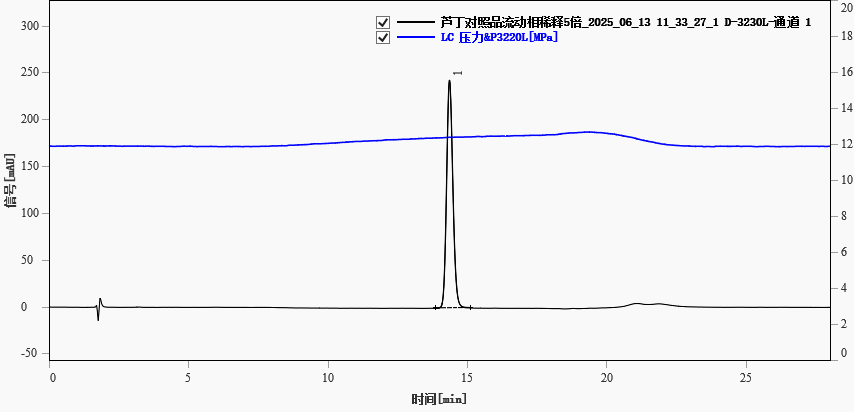
Fig. 1. Chromatogram of standard rutin(5-fold dilution)
Table 2. Chromatographic parameters of rutin standard (diluted 5-fold)
| Retention Time (min) |
Peak Height (mAU) |
Peak Area (mAU·s) |
Plate Number (N) |
Asymmetry Factor |
| 14.356 |
243.172 |
3811.401 |
19288 |
1.233 |
Solvent Effects
When the rutin standard was dissolved in pure methanol and injected without dilution with mobile phase, severe peak fronting was observed (asymmetry factor 0.730, plate number 6476). Diluting the sample with initial mobile phase effectively eliminated the solvent effect and improved peak symmetry.
Sample Analysis
The mulberry leaf sample solution was analyzed under the same conditions. The rutin peak eluted at approximately 14.380 min (Figure 2). Chromatographic parameters are given in Table 3.
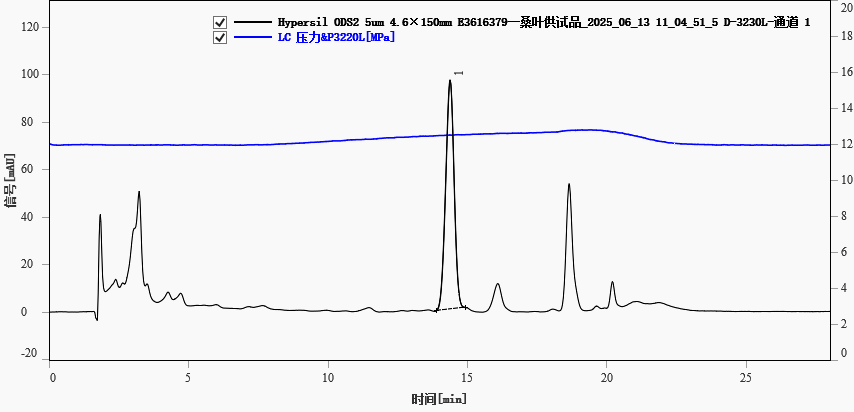
Fig. 2. Chromatogram of a mulberry leaf sample, peak 1-rutin
Table 3. Chromatographic parameters of rutin in mulberry leaf sample
| Retention Time (min) |
Peak Height (mAU) |
Peak Area (mAU·s) |
Plate Number (N) |
Asymmetry Factor |
| 14.380 |
96.333 |
2032.939 |
10521 |
0.905 |


