HPLC Determination of Enrofloxacin and Ciprofloxacin Lactate in Veterinary Drugs
- Analyte: Enrofloxacin, Ciprofloxacin Lactate
- System: Agress 1100 or EClassical 3100 HPLC System with UV Detector
- Column: Supersil ODS2 (5 μm, 4.6 × 150 mm)
- Highlight: Reliable HPLC methods for fluoroquinolone analysis in veterinary drug formulations
Introduction
Fluoroquinolones are a class of synthetic antibiotics widely used in livestock and aquaculture for the treatment of bacterial infections. They exert their antibacterial effect by inhibiting DNA replication enzymes such as DNA gyrase and topoisomerase IV, showing broad-spectrum activity against Gram-negative and some Gram-positive bacteria. Due to their extensive use, monitoring their concentration in veterinary drug formulations is essential for quality control and to prevent residues in animal-derived foods. High-performance liquid chromatography (HPLC) is a mainstream technique for fluoroquinolone analysis, typically employing reversed-phase C18 columns with UV or fluorescence detection. This method offers high sensitivity, good reproducibility, and compliance with national standards such as GB/T 20366-2006 and the Chinese Veterinary Pharmacopoeia.
Standards and Reagents
Standard: Ciprofloxacin lactate reference standard, enrofloxacin reference standard (used as internal standard)
Reagents: 0.025 mol/L phosphoric acid solution, acetonitrile (HPLC grade), triethylamine (for pH adjustment), 0.025 mol/L phosphoric acid solution/acetonitrile = 87:13 (v/v), adjusted to pH 3.0 with triethylamine (mobile phase)
Standard Solution Preparation
Ciprofloxacin lactate stock solution: Accurately weigh about 25 mg of ciprofloxacin lactate reference standard into a 100 mL volumetric flask. Dissolve and dilute to volume with mobile phase. Mix well.
Enrofloxacin internal standard solution: Accurately weigh 25 mg of enrofloxacin reference standard into a 100 mL volumetric flask. Dissolve and dilute to volume with mobile phase. Mix well.
Test solution: Precisely pipette 5 mL of ciprofloxacin lactate stock solution and 5 mL of enrofloxacin internal standard solution into a 25 mL volumetric flask. Dilute to volume with mobile phase and mix well.
Sample Pretreatment
For veterinary drug formulations containing ciprofloxacin lactate, prepare the sample solution following a similar procedure as the standard preparation, ensuring the final concentration is within the calibration range. Filter through a 0.45 μm membrane before injection.
Instruments and Equipment
HPLC System: Agress 1100 or EClassical 3100 equipped with UV-vis detector, high-pressure pump, manual injector (Rheodyne 7725i) or autosampler, column oven, Kromstation CDS.
Pretreatment equipment: Analytical balance, volumetric flasks, pipettes, membrane filtration apparatus, etc.
Chromatographic Conditions
- Column: Supersil ODS2 (5 μm, 4.6 × 250 mm)
- Mobile Phase: 0.025 mol/L phosphoric acid solution / acetonitrile = 87:13 (v/v), adjusted to pH 3.0 with triethylamine
- Flow Rate: 1.0 mL/min
- Detection: UV at 277 nm
- Injection Vol.: 20 μL
- Column Temp.: Ambient
Standard Separation
A typical chromatogram of ciprofloxacin lactate and enrofloxacin is shown in Figure 1. The two compounds are well resolved with retention times of approximately 10.52 min for ciprofloxacin lactate and 15.95 min for enrofloxacin. Chromatographic parameters are summarized in Table 1.
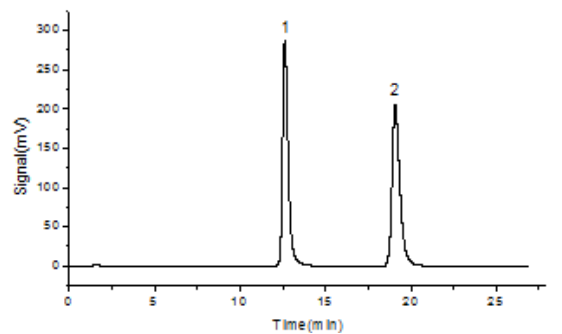
Fig. 1. Chromatogram of ciprofloxacin lactate (peak 1) and enrofloxacin(peak 2)
Table 1. Chromatographic parameters for ciprofloxacin lactate and enrofloxacin
| Compound |
Retention Time (min) |
Tailing Factor |
Plate Number (N) |
| Ciprofloxacin Lactate |
10.52 |
1.36 |
8400 |
| Enrofloxacin |
15.95 |
1.28 |
- |

