Cefoperazone is a third-generation cephalosporin antibiotic, and sulbactam is an irreversible β-lactamase inhibitor that enhances the activity of β-lactam antibiotics. Their combination is widely used to treat various bacterial infections. According to the pharmacopoeial method for cefoperazone sodium and sulbactam sodium, proper separation of the two components and related substances is essential for quality control. This report presents an HPLC analysis of cefoperazone and sulbactam reference standards using an iChrom5100 system, addressing an issue where a degradation peak of cefoperazone was not observed with a new column but appeared with an older column of the same type. The method verification was performed to ensure system suitability and peak resolution.
Standards: cefoperazone reference standard and sulbactam reference standard (provided by client)
Reagents: deionized water (18.2 MΩ·cm), acetonitrile (HPLC grade), Tetrabutylammonium hydroxide (55% solution, AR), phosphoric acid (AR, for pH adjustment), sodium dihydrogen phosphate (AR), disodium hydrogen phosphate (AR), other solvents and chemicals as required for sample pretreatment (e.g., extraction solvents, filtration membranes)
Standard Solution Preparation
Phosphate buffer (0.2 mol/L): Dissolve 1.217 g of sodium dihydrogen phosphate and 4.369 g of disodium hydrogen phosphate in water, dilute to 100 mL in a volumetric flask.
Tetrabutylammonium hydroxide solution (0.005 mol/L, pH 4.0): Pipette 2.4 mL of 55% tetrabutylammonium hydroxide solution into a 1 L volumetric flask, add about 900 mL of water, adjust pH to 4.0 with phosphoric acid, and dilute to volume.
Cefoperazone test solution (2 mg/mL): Accurately weigh about 20.4 mg of cefoperazone sample into a 10 mL volumetric flask, add 1 mL of phosphate buffer to dissolve, then dilute to volume with the mobile phase.
Sulbactam test solution (2 mg/mL): Accurately weigh about 20.9 mg of sulbactam sample into a 10 mL volumetric flask, add 1 mL of phosphate buffer to dissolve, then dilute to volume with the mobile phase.
Mixed test solution (1 mg/mL each): Pipette 0.5 mL each of cefoperazone and sulbactam test solutions into a vial and mix well.
All solutions are filtered through a 0.45 μm membrane before injection
No further pretreatment is required beyond dissolution and filtration.
HPLC System: EClassical 3200 equipped with quaternary pump, UV-vis detector, autosampler, column oven, solvent bottle tray, Kromstation CDS.
Pretreatment equipment: analytical balance (0.0001 g sensitivity), ultrasonic cleaner, centrifuge, solvent filtration apparatus, membrane filters, pipettes, etc.
Column: Hypersil ODS2 (5μm, 4.6 × 150 mm)
Mobile Phase: Acetonitrile / 0.005 mol/L tetrabutylammonium hydroxide solution (pH adjusted to 3.98) = 25 / 75 (v/v)
Flow Rate: 1.0 mL/min
Detection: UV at 220 nm
Injection Vol.: 10 μL
Column Temp.: 30°C
The mixed test solution (cefoperazone and sulbactam) was analyzed under the above conditions. A representative chromatogram is shown in Figure 1. Five main peaks were observed with retention times of approximately 3.544, 5.160, 5.780, 6.876, and 11.606 min. The critical pair for method suitability is the peaks at 5.160 min and 5.780 min, which correspond to a cefoperazone-related substance and sulbactam, respectively. Their resolution was calculated as 2.866, well above the required 1.5. The sulbactam peak shape is acceptable (asymmetry 1.937), and the cefoperazone main peak shows good symmetry. Chromatographic parameters are summarized in Table 1.
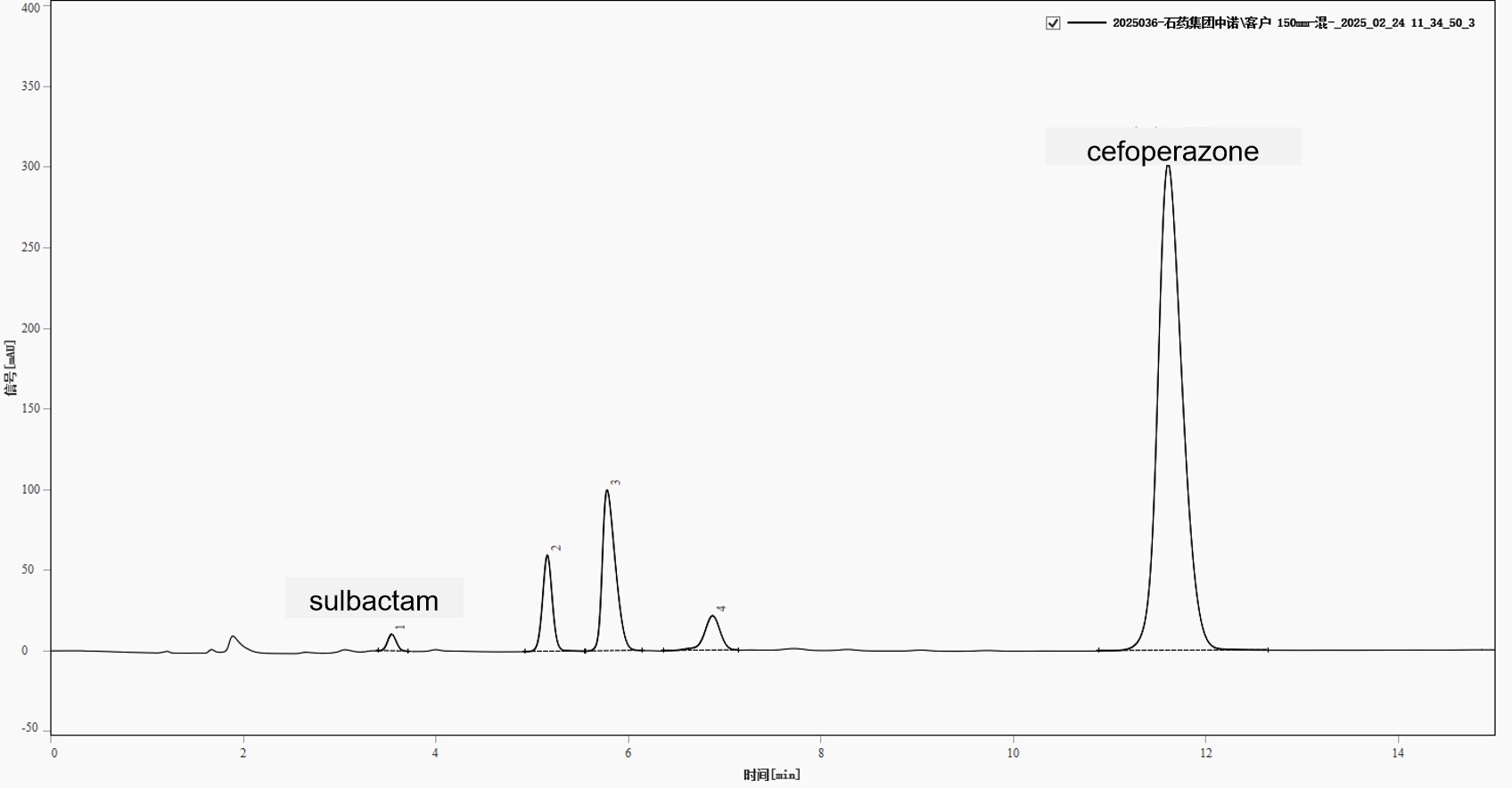
Fig. 1. Chromatogram of mixed test solution (cefoperazone and sulbactam)
Table 1. Chromatographic parameters of cefoperazone and sulbactam mixed test solution

