Determination of Synthetic Colorants in Meat Products
- Analyte: Tartrazine, Amaranth, Ponceau 4R, Sunset Yellow, Allura Red
- System: EClassical 3200 HPLC System with UV-Vis Detector
- Column: Supersil ODS2 (5 μm, 4.6 × 250 mm)
- Highlight: Simultaneous determination of five synthetic colorants in meat products with excellent linearity and sensitivity.
Introduction
Synthetic colorants are widely used in food processing to improve appearance. However, their misuse can mask inferior quality and pose health risks. In meat products, certain colorants like tartrazine, amaranth, ponceau 4R, and sunset yellow are prohibited, while allura red is restricted to specific products. This method, based on GB/T 9695.6-2008 and GB 2760-2014, uses HPLC for the simultaneous determination of five synthetic colorants in meat products, validating linearity, recovery, and real-sample applicability.
Standards and Reagents
Standards: Tartrazine, Amaranth, Ponceau 4R, Sunset Yellow, Allura Red
Reagents: Methanol (HPLC grade), Ammonium acetate (analytical grade), Purified water (18.2 MΩ·cm), Petroleum ether (analytical grade), Ammonia solution (analytical grade), Formic acid (analytical grade), Anhydrous ethanol (analytical grade), SPE cartridge: Polyamide PA-1 (500 mg/6 mL).
Standard Solution Preparation
Ammonium acetate solution (0.02 mol/L): Dissolve 1.54 g of ammonium acetate in 1000 mL water and filter through a 0.45 μm membrane.
Methanol-formic acid solution: Mix 60 mL methanol and 40 mL formic acid.
Ethanol-ammonia solution: Mix 70 mL anhydrous ethanol, 20 mL ammonia solution, and 10 mL water.
5% Ammoniated methanol: Dilute 5 mL ammonia solution to 100 mL with methanol.
Mixed standard stock solution (100 μg/mL): Accurately weigh 10 mg each of tartrazine, amaranth, ponceau 4R, sunset yellow, and allura red into a 100 mL volumetric flask, dissolve and dilute to volume with water.
Mixed standard working solutions: Dilute the stock solution with mobile phase to obtain concentrations of 1, 5, 10, 20, 40, 50, and 100 μg/mL.
Sample Pretreatment
- Sample homogenization: Mince and homogenize the meat sample.
- Defatting: Accurately weigh 2.0 g of homogenized sample into a 50 mL centrifuge tube, add 20 mL petroleum ether, vortex for 1 min, centrifuge at 5000 r/min for 5 min, and discard the supernatant. Repeat once.
- Extraction: Remove residual petroleum ether under nitrogen. Add 5 mL ethanol-ammonia solution, vortex for 1 min, sonicate for 30 min, centrifuge at 10,000 r/min for 5 min, and collect the supernatant. Repeat extraction once and combine the supernatants.
- Concentration: Evaporate the combined supernatant under nitrogen at 75°C to about 2 mL, transfer to a 10 mL centrifuge tube, and adjust pH to approximately 6 with citric acid.
SPE Purification
- Activation: Condition the polyamide PA-1 cartridge with 5 mL methanol and 5 mL water.
- Loading: Pass the sample solution through the cartridge.
- Washing: Wash with 2 mL water, then 2 mL methanol-formic acid solution, and dry under vacuum.
- Elution: Elute with 5 mL 5% ammoniated methanol, collect the eluate, and evaporate to dryness under nitrogen at 65°C.
- Reconstitution: Dissolve the residue in 1 mL mobile phase, filter through a 0.45 μm membrane, and inject 10 μL into the HPLC system.
Instruments and Equipment
HPLC System (EClassical 3200):D3210 UV-Vis detector, P3220 high-pressure pump, S3210 autosampler, O3220 column oven, T3200 solvent bottle tray, Kromstation CDS.
Pretreatment Equipment: Vortex mixer, analytical balance, homogenizer, centrifuge, nitrogen evaporator, solid-phase extraction manifold, volumetric flasks, pipettes, etc.
Chromatographic Conditions
Mobile Phase: A: 0.02 mol/L ammonium acetate solution; B: Methanol, in gradient (Table 1)
Table 1. Gradient Program
| Time (min) |
A% |
B% |
| 0 |
80 |
20 |
| 6 |
65 |
35 |
| 15 |
40 |
60 |
| 20 |
80 |
20 |
| 30 |
80 |
20 |
Flow Rate: 1.0 mL/min
Column Temperature: 35°C
Injection Volume: 10 μL
Detection: Detection at different wavelength (Table 2)
Table 2. Detection wavelength program
| Time (min) |
Wavelength (nm) |
| 0.0 |
428 |
| 6.7 |
428 |
| 6.7 |
255 |
| 9.2 |
255 |
| 9.2 |
500 |
| 11.7 |
500 |
| 11.7 |
507 |
| 14.2 |
507 |
Experimental Results
Standard Separation
A chromatogram of a mixed standard solution (10 μg/mL) is shown in Figure 1. Chromatographic parameters are summarized in Table 3.
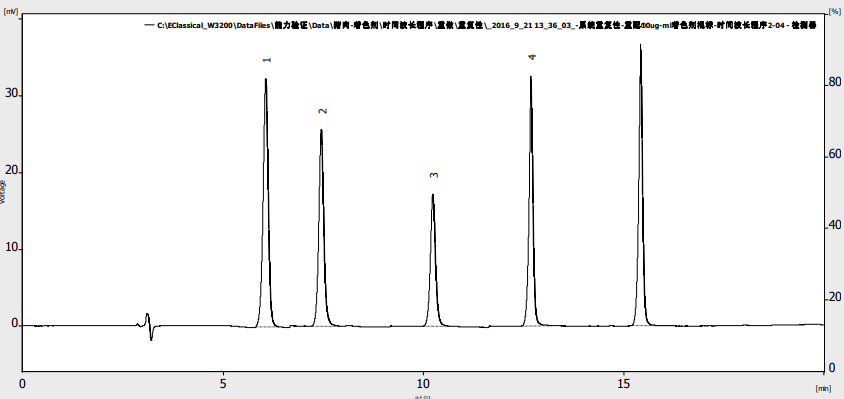
Fig. 1. Chromatogram of separation for synthetic artificial pigment mixture standard (10 µg/mL).
Peak, 1-citron yellow, 2-amaranth red, 3-carmine, 4-sunset yellow, 5-indigo
Table 3. Chromatographic parameters for colorant standards
| Compound |
Retention Time (min) |
Peak Height (mAU) |
Tailing Factor |
Resolution |
| Tartrazine |
6.082 |
232.289 |
0.966 |
--- |
| Amaranth |
7.468 |
25.660 |
1.071 |
6.310 |
| Ponceau 4R |
10.253 |
17.228 |
1.079 |
12.887 |
| Sunset Yellow |
12.705 |
32.520 |
0.962 |
12.016 |
| Allura Red |
15.443 |
36.736 |
0.964 |
14.573 |
Method Repeatability
Seven consecutive injections of a 10.0 μg/mL mixed standard solution were performed. Retention time RSD values are shown in Table 4, indicating excellent system precision.
Table 4. System repeatability (n=7)
| Injection |
Tartrazine (min) |
Amaranth (min) |
Ponceau 4R (min) |
Sunset Yellow (min) |
Allura Red (min) |
| 1 |
6.075 |
7.463 |
10.251 |
12.703 |
15.442 |
| 2 |
6.092 |
7.479 |
10.269 |
12.723 |
15.466 |
| 3 |
6.099 |
7.485 |
10.277 |
12.731 |
15.468 |
| 4 |
6.082 |
7.468 |
10.253 |
12.705 |
15.443 |
| 5 |
6.074 |
7.461 |
10.246 |
12.699 |
15.446 |
| 6 |
6.088 |
7.481 |
10.276 |
12.733 |
15.474 |
| 7 |
6.067 |
7.458 |
10.249 |
12.702 |
15.443 |
| Mean |
6.082 |
7.471 |
10.260 |
12.714 |
15.453 |
| RSD% |
0.19 |
0.14 |
0.13 |
0.12 |
0.10 |
Linearity and Detection Limit
Calibration curves were constructed using mixed standard solutions at concentrations of 0.2, 0.4, 0.8, 2.0, 4.0, 8.0, and 20.0 μg/mL. Linear equations and correlation coefficients are given in Table 5. Instrument and method detection limits are listed in Table 6.
Table 5. Linearity Parameters for Colorant Standards
| Compound |
Linear Range (μg/mL) |
Correlation Coefficient (R) |
Linear Equation |
| Tartrazine |
0.2–20 |
1.0000 |
y = 31.8441x – 0.4752 |
| Amaranth |
0.2–20 |
1.0000 |
y = 25.5906x – 0.6254 |
| Ponceau 4R |
0.2–20 |
1.0000 |
y = 17.7863x + 0.2739 |
| Sunset Yellow |
0.2–20 |
1.0000 |
y = 28.0900x – 0.1074 |
| Allura Red |
0.2–20 |
0.9999 |
y = 33.1146x + 0.7895 |
Table 6. Detection Limits
| Compound |
Instrument LOD (ng/mL) |
Method LOD (μg/kg) |
| Tartrazine |
20.0 |
12.5 |
| Amaranth |
22.0 |
13.8 |
| Ponceau 4R |
33.0 |
20.6 |
| Sunset Yellow |
20.0 |
12.5 |
| Allura Red |
17.0 |
10.6 |
Sample Analysis
The chromatogram of the analysis of colorants in several meat products is shown in Figure 2. Recovery was evaluated by spiking blank pork samples at two concentration levels (5 μg/mL and 10 μg/mL). Results are shown in Table 7.
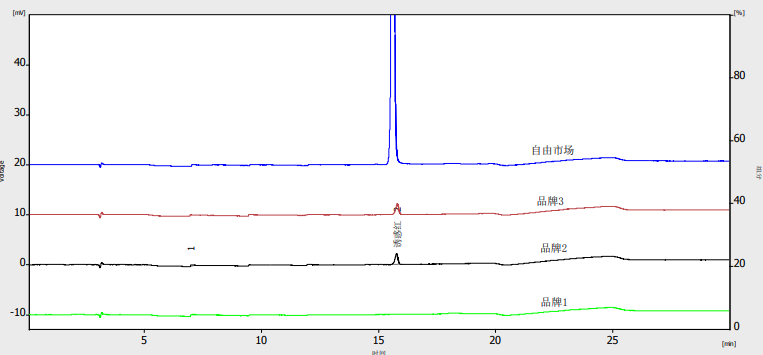
Fig. 2. Chromatogram of the analysis of colorants in several meat products
Peak, 1-citron yellow, 2-amaranth red, 3-carmine, 4-sunset yellow, 5-indigo
Table 7. Spike recovery results
| Spike Level |
Sample |
Tartrazine |
Amaranth |
Ponceau 4R |
Sunset Yellow |
Allura Red |
| 5 μg/mL |
Peak Area (mAU·s) |
66.288 |
46.916 |
33.491 |
56.045 |
67.897 |
| Recovery (%) |
91.4 |
87.5 |
92.5 |
90.3 |
105.9 |
| 10 μg/mL |
Peak Area (mAU·s) |
113.604 |
90.941 |
67.688 |
120.562 |
134.535 |
| Recovery (%) |
79.7 |
83.6 |
89.9 |
98.9 |
98.5 |
Several commercial meat products were analyzed. The results are summarized in Table 8.
Table 8. Colorant content in meat products (μg/g)
| Sample |
Tartrazine |
Amaranth |
Ponceau 4R |
Sunset Yellow |
Allura Red |
| Brand 1 – Luncheon Meat |
0 |
0 |
0 |
0 |
0 |
| Brand 2 – Bone-in Ham |
0 |
0 |
0 |
0.80 |
0 |
| Brand 3 – Smoked Ham |
0 |
0 |
0 |
0.74 |
0 |
| Free Market – Sausage |
0 |
0 |
0 |
30.56 |
0 |
References
[1] GB 5009.35-2016 National Food Safety Standard – Determination of Synthetic Colorants in Foods
[2] GB/T 9695.6-2008 Determination of Ponceau 4R in Meat Products
[3] GB 2760-2014 National Food Safety Standard – Standard for Use of Food Additives


