Determination of Benzoic Acid and Sorbic Acid in Sparkling Beverage
- Analyte: Benzoic Acid, Sorbic Acid
- System: EClassical 3200 HPLC
- Column: Supersil AQ-C18 (5μm, 4.6×250mm)
- Highlight: Detection of two preservatives in sparkling beverage with good resolution and linearity.
Introduction
Benzoic acid and sorbic acid are commonly used synthetic preservatives. Benzoic acid is inexpensive but slightly more toxic, while potassium sorbate has strong antibacterial activity and low toxicity. This protocol determines benzoic acid and sorbic acid in food using High Performance Liquid Chromatography (HPLC) according to the national standard GB5009.28-2016.
Standards and Reagents
Standards: Benzoic acid (analytical grade), Sorbic acid (content ≥98%)
Reagents: Methanol (chromatographic grade), Ammonium acetate (analytical grade), Deionized water (18.2MΩ)
Standard Solution Preparation
Stock Solutions: Accurately weigh 0.1g of benzoic acid and sorbic acid separately, dissolve and dilute to 100mL with methanol to obtain 1mg/mL single standard stock solutions.
Mixed Standard Solution: Pipette 10mL of each stock solution into a 50mL volumetric flask and dilute to volume with methanol to obtain a 0.2mg/mL mixed standard solution.
Series Working Solutions: Dilute the mixed standard solution to concentrations of 2, 5, 10, 20, 50, 100, 200 mg/L.
Sample Pretreatment
Accurately weigh 2g of the sample (to 0.001g) into a 50mL centrifuge tube. Add approximately 25mL of water, vortex mix, and sonicate in a 50°C water bath for 20min. Cool to room temperature, then centrifuge at 8000r/min for 5min. Transfer the aqueous phase to a 50mL volumetric flask, dilute to volume with water, mix well, and filter through a 0.45μm membrane filter before analysis.
Instruments and Equipment
Liquid Chromatography System: EClassical 3200 (including P3200 high-pressure constant flow pump, D3200 UV-Vis detector, S3200 autosampler, O3220 column oven, etc.)
Pretreatment Equipment: Solvent filter apparatus, diaphragm vacuum pump, ultrasonic cleaner, precision electronic balance, vortex mixer, centrifuge, volumetric flasks, micropipettes, etc.
Chromatographic Conditions
- Column: Supersil AQ-C18 (5μm, 4.6×250mm)
- Mobile Phase: Methanol / 0.02M Ammonium acetate solution = 5 / 95 (V/V)
- Flow Rate: 1.0 mL/min
- Detection: UV at 230 nm
- Injection Volume: 10 μL
- Column Temp.: Ambient
Experimental Results
Standard Separation
Retention time for benzoic acid was 10.90 min, for sorbic acid 14.93 min, with good resolution (Figure 1).
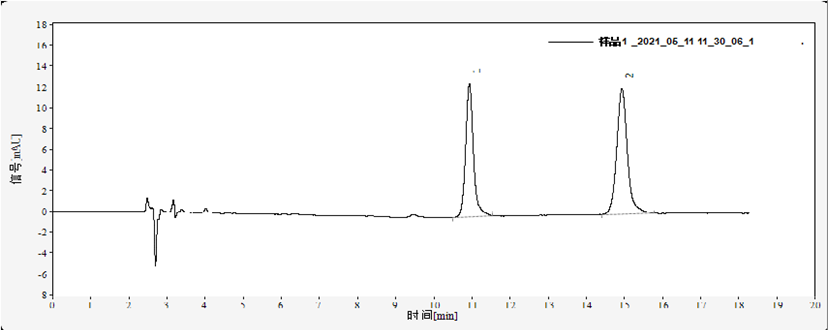
Fig. 1. Chromatogram of 20 mg/L mixed standard solution of benzoic acid and sorbic acid
Linearity
Within 2~200mg/L, linear equation y=35.552x+13.847 for benzoic acid, R=0.9997; y=68.224x-48.443, R=0.9995 for sorbic acid:
Sample Analysis
Sample analysis results of a commercial sparkling beverage was shown in Figure 2 and Table 1, the content of benzoic acid was 0.1165 g/kg, and sorbic acid was 0.1035 g/kg.
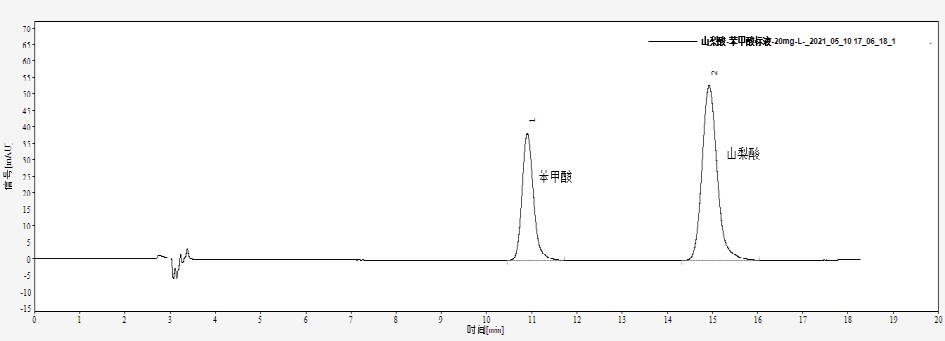
Fig. 2. Chromatogram of benzoic acid and sorbic acid in carbonated beverages
Table 1. Sample analysis results
| Sample |
Retention Time [min] |
Peak Area [mAU·s] |
Plate Number [N/m] |
Tailing Factor |
Content (g/kg) |
| Benzoic Acid |
10.93 |
179.51 |
63500 |
1.16 |
0.1165 |
| Sorbic Acid |
14.94 |
234.11 |
61600 |
1.13 |
0.1035 |


