Determination of Amoxicillin Sodium and Clavulanate Potassium by HPLC
- Analyte: Amoxicillin Sodium and Clavulanate Potassium
- System: EClassical 3200 HPLC with UV Detector
- Column: Sinopak BEH AQ (5 µm, 4.6×250 mm)
- Highlight: A reliable HPLC method for amoxicillin sodium and clavulanate potassium meeting ChP requirements with good peak symmetry and resolution
Introduction
Amoxicillin sodium is a broad-spectrum penicillin antibiotic, while clavulanate potassium is a β-lactamase inhibitor that protects amoxicillin from enzymatic degradation. Their combination is widely used in pharmaceutical preparations. According to the 2025 Chinese Pharmacopoeia (draft), the system suitability test for HPLC analysis requires tailing factors between 0.8 and 1.8 for quantitative analysis using peak area. This report presents an HPLC method for the determination of amoxicillin sodium and clavulanate potassium using an Elite 3200 system with UV detection, compliant with pharmacopoeial requirements.
Reagents and Test Sample
Reagents: phosphoric acid (AR), sodium dihydrogen phosphate (AR), methanol (HPLC grade), deionized water (18.2 MΩ·cm), other solvents and chemicals as required for sample pretreatment (e.g., extraction solvents, filtration membranes)
Test sample: the amoxicillin sodium and clavulanate potassium for injection (provided by the client)
System suitability solution: Accurately weigh 0.0084 g of the test sample into a 10 mL volumetric flask, dissolve and dilute to volume with the mobile phase.
Sample solution: Accurately weigh 0.0112 g of the test sample into a 10 mL volumetric flask, dissolve and dilute to volume with purified water.
All solutions are filtered through a 0.45 µm membrane before injection.
Instruments and Equipment
HPLC System: EClassical 3200 equipped with quaternary pump, UV-vis detector, autosampler, column oven, solvent bottle tray, Kromstation CDS.
Pretreatment equipment: analytical balance (0.0001 g sensitivity), ultrasonic cleaner, centrifuge, solvent filtration apparatus, membrane filters, pipettes, etc.
Chromatographic Conditions
Column: Sinopak BEH AQ (5 µm, 4.6×250 mm)
Mobile Phase: 0.05 mol/L sodium dihydrogen phosphate solution (pH adjusted to 4.0 with phosphoric acid) / methanol = 95 / 5 (v/v)
Flow Rate: 1.0 mL/min
Detection: UV at 220 nm
Injection Vol.: 20 μL
Column Temp.: 30°C
Experimental Results
System suitability solution separation
A typical chromatogram of the system suitability solution is shown in Figure 1. Four main peaks are well resolved with retention times of approximately 5.845, 7.042, 8.006, and 9.139 min. Chromatographic parameters are summarized in Table 1, all meeting the ChP equirements (tailing factor 0.8–1.8, resolution >1.5).
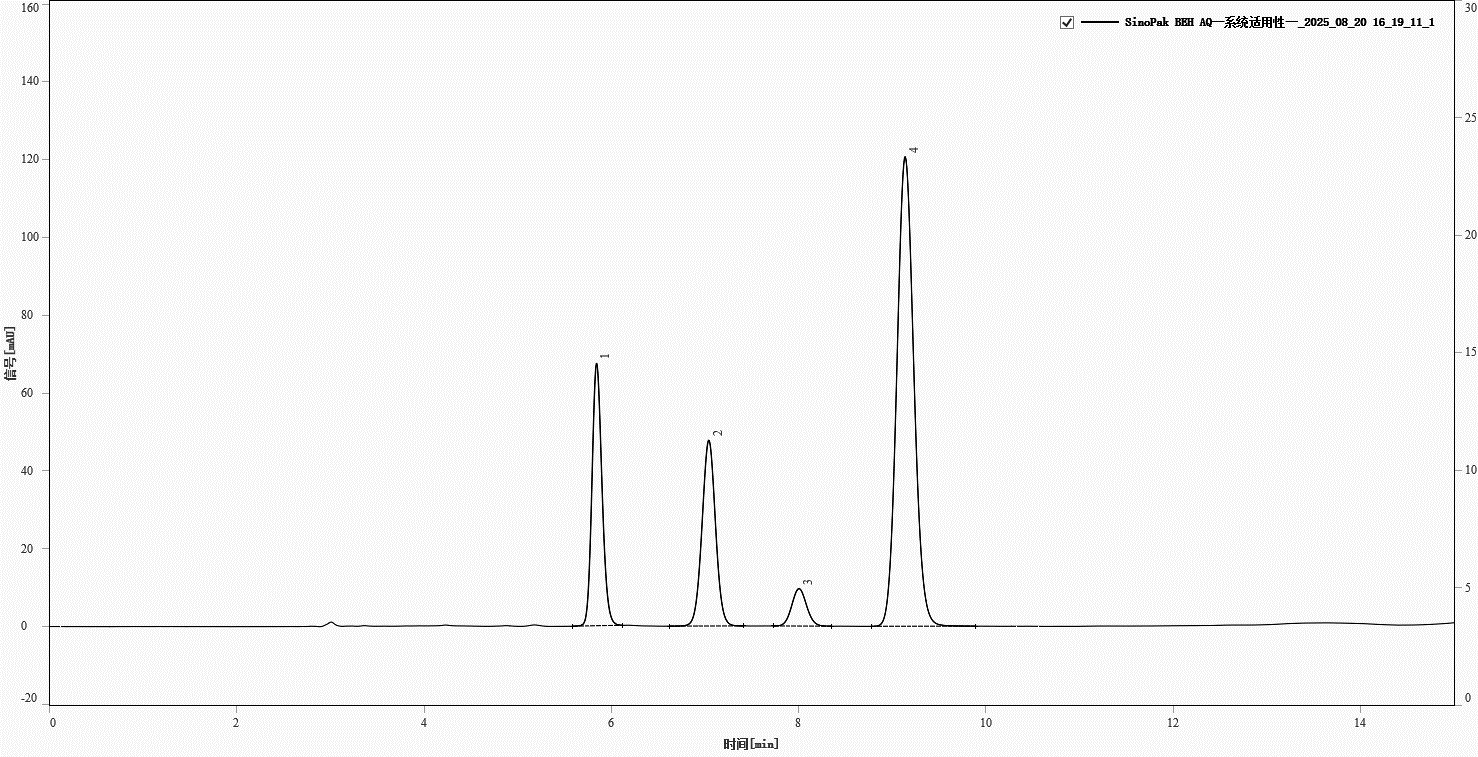
Fig. 1. Chromatogram of system suitability solution. Peaks, 1-amoxicillin, 2-clavulanate, 3-and 4-impurities
Table 1. Chromatographic parameters of system suitability solution
| Peak No. |
Retention Time (min) |
Peak Area (mAU·s) |
Tailing Factor |
Plate Number (N) |
Resolution |
| 1 |
5.845 |
502.231 |
1.263 |
14311 |
– |
| 2 |
7.042 |
479.725 |
1.023 |
11560 |
5.237 |
| 3 |
8.006 |
105.851 |
1.117 |
12050 |
3.480 |
| 4 |
9.139 |
1523.332 |
1.127 |
12274 |
3.647 |
Method Performance
Method Repeatability
The sample solution was injected ten times consecutively (Figure 2). The retention time RSDs for the four peaks were below 0.33%, indicating excellent system precision. Note that peaks 2 and 3 (retention times ~6.95 and 7.99 min) showed increasing peak areas over time (possibly due to instability), so their area RSDs were not calculated. Detailed data are given in Table 2.
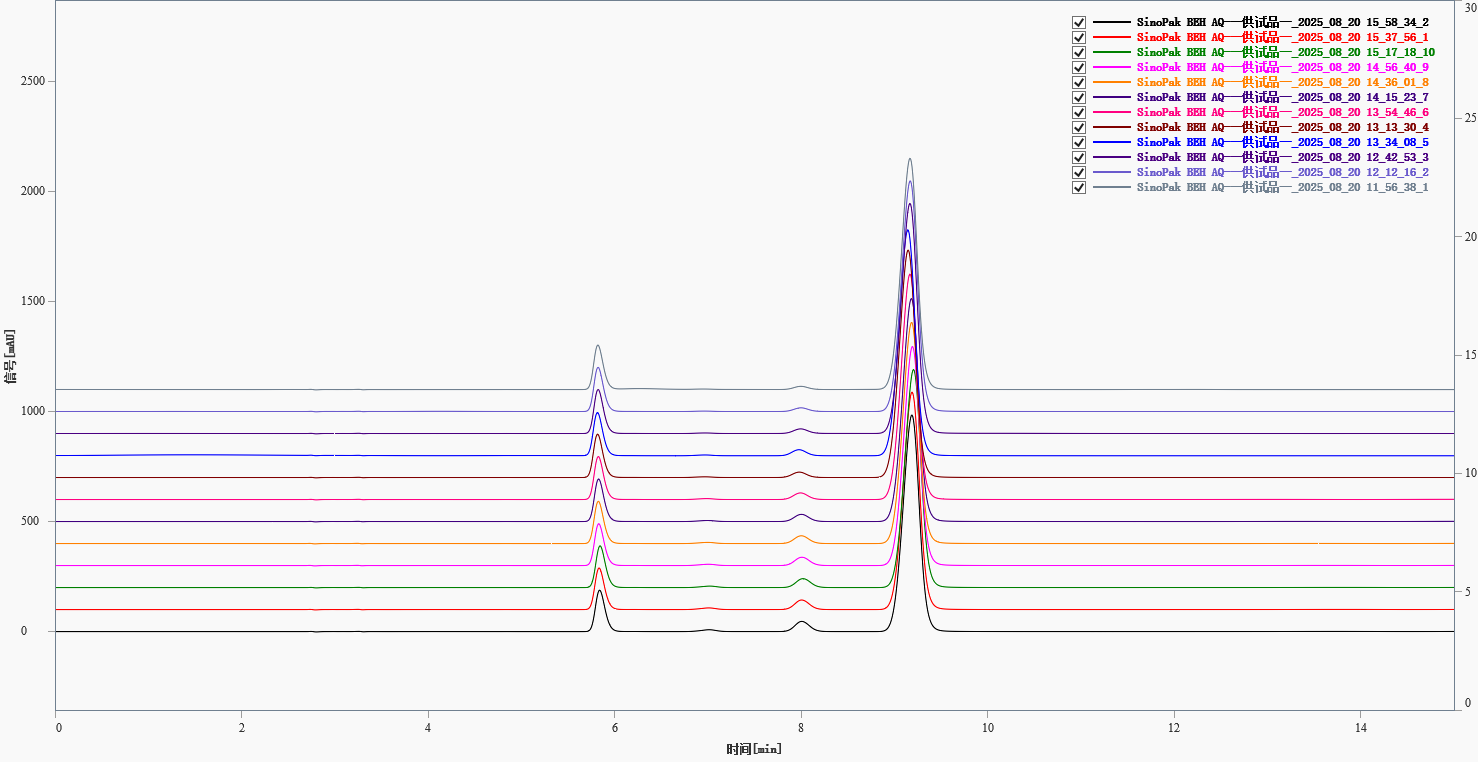
Fig. 2. Overlay chromatogram of 10 replicate sample injections.
Table 2. Repeatability of sample analysis (10 injections)
| Peak No. |
Retention Time RSD (%) |
Area RSD (%) |
Tailing Factor Range |
Plate Number Range |
| 1 |
0.15 |
– |
1.21–1.26 |
14409–14814 |
| 2 |
0.33 |
– |
0.91–1.04 |
6279–7856 |
| 3 |
0.15 |
– |
1.04–1.07 |
11360–37878 |
| 4 |
0.20 |
– |
0.93–0.94 |
12181–12456 |
Conclusion
The results demonstrate that the Elite 3200 HPLC system equipped with a Sinopak BEH AQ column meets the system suitability requirements of the Chinese Pharmacopoeia for the analysis of amoxicillin sodium and clavulanate potassium. All peaks exhibit tailing factors within 0.8–1.8 and resolutions >1.5. The method shows good retention time precision (RSD <0.33%).


