Determination of 16 PAHs in Environmental Samples by HPLC
- Analyte: 16 PAHs
- System: EClassical 3200 HPLC System with UV Detector
- Column: Supersil ODS2 (5 μm, 4.6 × 250 mm)
- Highlight: A sensitive and reliable HPLC method for simultaneous determination of 16 PAHs in water, air, and soil, meeting multiple environmental standards.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are a large class of organic compounds containing two or more fused benzene rings. They are potent carcinogens and are widely distributed in the environment due to incomplete combustion of fossil fuels, wood, and other organic materials. PAHs pose serious risks to human health, including carcinogenicity, mutagenicity, and teratogenicity. In recent years, increased human activities have disrupted the natural balance of PAHs, leading to elevated environmental levels. Therefore, monitoring PAHs in water, air, and soil is crucial. This method is based on several Chinese standards, including HJ 478-2009 (water), HJ 647-2013 (air), and HJ 784-2016 (soil and sediment), and provides a complete HPLC solution for the analysis of 16 priority PAHs (Naphthalene, Acenaphthylene, Fluorene, Acenaphthene, Phenanthrene, Anthracene, Fluoranthene, Pyrene, Chrysene, Benzo[a]anthracene, Benzo[b]fluoranthene, Benzo[k]fluoranthene, Benzo[a]pyrene, Dibenzo[a,h]anthracene, Benzo[ghi]perylene, Indeno[1,2,3-cd]pyrene).
Standards and Reagents
Standards
PAH standard mixture: 16 PAHs certified reference material (from National Standard Materials Network), 200 μg/mL in acetonitrile.
Decafluorobiphenyl (optional internal standard, from J&K Scientific).
Reagents
Acetonitrile, HPLC grade; Deionized Water, ≥18.2 MΩ·cm.
Other materials
Volumetric flasks, pipettes, membrane filters (0.45 μm), etc.
Standard Solution Preparation
Mixed stock solution (20 μg/mL): Accurately pipette 1 mL of the 200 μg/mL PAH standard into a 10 mL volumetric flask and dilute to volume with acetonitrile.
Working standard solutions: Dilute the stock solution with acetonitrile to obtain concentrations of 0.1, 0.5, 1.0, 5.0, and 10.0 μg/mL.
Optional internal standard solution: Prepare a solution containing 5.0 μg/mL of 16 PAHs and 2.0 μg/mL of decafluorobiphenyl in acetonitrile.
Sample Pretreatment
Sample preparation follows the procedures described in HJ 478-2009, HJ 647-2013, and HJ 784-2016. General steps include:
Water samples
Extract with liquid-liquid extraction (e.g., cyclohexane) or solid-phase extraction (e.g., C18 cartridge). Concentrate the extract and redissolve in acetonitrile.
Air samples
Collect particulate matter and gaseous phase on filters and adsorbents, then extract with appropriate solvents (e.g., acetonitrile) by sonication or Soxhlet extraction.
Soil/sediment samples
Extract by Soxhlet or pressurized fluid extraction with dichloromethane/acetone, concentrate, and perform cleanup if necessary.
All final extracts are filtered through a 0.45 μm membrane before HPLC analysis.
Instruments and Equipment
HPLC System Options
Agress 1100: Two P1100 pumps, D1100 UV detector, O1100 column oven, Rheodyne 7725i injector, TD-1-15 gradient mixer, chromatography data station.
EClassical 3200: P3220 pump, D3210 UV detector, O3220 column oven, Rheodyne 7725i injector, gradient elution capability.
Pretreatment equipment
Solvent filtration apparatus, vacuum pump, ultrasonic cleaner, rotary evaporator, nitrogen evaporator, etc.
Chromatographic Conditions
Column: Supersil ODS2 (5 μm, 4.6 × 250 mm)
Mobile phase: A: Acetonitrile; B: Water, in gradient (Table 1).
Table 1. Gradient program
| Time (min) |
A% |
B% |
| 0 |
70 |
30 |
| 16 |
70 |
30 |
| 40 |
100 |
0 |
| 45 |
100 |
0 |
Flow rate: 1.0 mL/min
Detection wavelength: 220 nm (or variable; 254 nm also common)
Injection volume: 10 μL
Column temperature: 30°C
Experimental Discussion and Results
Temperature Optimization
The effect of column temperature on separation was studied at 25, 30, 35, and 40°C (Figure 1). As temperature increased, resolution for critical pairs (e.g., chrysene/benzo[a]anthracene and benzo[ghi]perylene/indeno[1,2,3-cd]pyrene) decreased. A temperature of 30°C was selected as optimal for baseline separation of all 16 PAHs.
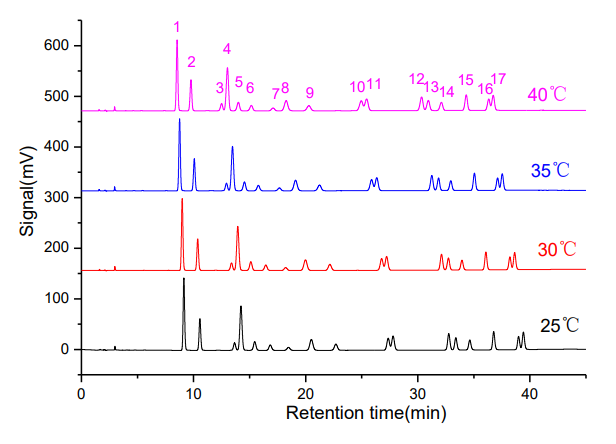
Figure 1-1. Overlay chromatograms of 16 PAHs at different temperatures (25, 30, 35, 40°C)
Typical Chromatogram
A standard solution containing 5.0 μg/mL of 16 PAHs and 2.0 μg/mL of decafluorobiphenyl was analyzed at 30°C. The chromatogram (Figure 2) shows excellent separation of all compounds. Chromatographic parameters are summarized in Table 2.
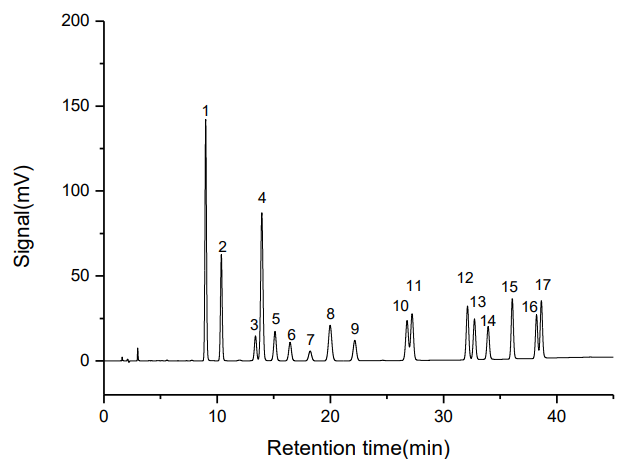
Figure 2. Typical chromatogram of 16 PAHs and decafluorobiphenyl standard (peak 7)
Table 2. Chromatographic parameters of 16 PAHs
| Peak |
Compound |
RT (min) |
Peak Area (mV·s) |
Asymmetry |
Plate Number (N/m) |
| 1 |
Naphthalene |
8.97 |
355.15 |
1.07 |
80900 |
| 2 |
Acenaphthylene |
10.35 |
673.25 |
1.05 |
84500 |
| 3 |
Fluorene |
13.36 |
204.85 |
1.04 |
85500 |
| 4 |
Acenaphthene |
13.91 |
1256.50 |
1.02 |
86600 |
| 5 |
Phenanthrene |
15.07 |
275.31 |
0.99 |
84400 |
| 6 |
Anthracene |
16.40 |
185.25 |
1.01 |
85400 |
| 7 |
Decafluorobiphenyl (IS) |
18.18 |
111.26 |
1.01 |
84600 |
| 8 |
Fluoranthene |
19.93 |
439.11 |
1.03 |
83300 |
| 9 |
Pyrene |
22.11 |
241.77 |
0.98 |
111500 |
| 10 |
Chrysene |
26.75 |
406.00 |
0.85 |
199800 |
| 11 |
Benzo[a]anthracene |
27.20 |
476.03 |
1.08 |
215400 |
| 12 |
Benzo[b]fluoranthene |
32.11 |
485.84 |
1.02 |
399400 |
| 13 |
Benzo[k]fluoranthene |
32.73 |
364.46 |
1.00 |
432800 |
| 14 |
Benzo[a]pyrene |
33.93 |
299.51 |
1.03 |
465300 |
| 15 |
Dibenzo[a,h]anthracene |
36.06 |
490.06 |
1.05 |
648300 |
| 16 |
Benzo[ghi]perylene |
38.20 |
351.88 |
1.04 |
699500 |
| 17 |
Indeno[1,2,3-cd]pyrene |
38.63 |
490.21 |
0.96 |
677900 |
Linearity
Working standard solutions at 0.1, 0.5, 1.0, 5.0, and 10.0 μg/mL were injected. Calibration curves were constructed by plotting peak area against concentration. Linear equations and correlation coefficients are given in Table 3. All compounds show excellent linearity (R ≥ 0.999) over the range of 0.1–10 μg/mL.
Table 3. Linear equations for 16 PAHs
| Peak |
Compound |
Linear Equation |
R |
| 1 |
Naphthalene |
y = 281.62x + 15.76 |
0.9996 |
| 2 |
Acenaphthylene |
y = 120.73x + 0.635 |
0.9999 |
| 3 |
Fluorene |
y = 43.252x – 0.687 |
0.9999 |
| 4 |
Acenaphthene |
y = 228.11x + 0.884 |
0.9999 |
| 5 |
Phenanthrene |
y = 56.573x – 0.788 |
0.9999 |
| 6 |
Anthracene |
y = 35.610x – 0.125 |
0.9999 |
| 8 |
Fluoranthene |
y = 85.312x – 2.013 |
0.9999 |
| 9 |
Pyrene |
y = 42.227x + 2.961 |
0.9997 |
| 10 |
Chrysene |
y = 79.638x – 0.977 |
0.9999 |
| 11 |
Benzo[a]anthracene |
y = 91.550x – 1.074 |
0.9999 |
| 12 |
Benzo[b]fluoranthene |
y = 93.395x – 0.736 |
0.9999 |
| 13 |
Benzo[k]fluoranthene |
y = 69.610x – 0.0072 |
0.9999 |
| 14 |
Benzo[a]pyrene |
y = 57.698x – 2.477 |
0.9998 |
| 15 |
Dibenzo[a,h]anthracene |
y = 96.439x – 0.105 |
0.9999 |
| 16 |
Benzo[ghi]perylene |
y = 68.375x – 0.981 |
0.9999 |
| 17 |
Indeno[1,2,3-cd]pyrene |
y = 94.666x – 0.585 |
0.9999 |
Detection Limits
Instrument detection limits (S/N = 3) were determined and converted to method detection limits (Table 4). The method achieves sub-ng/mL sensitivity, well below the requirements of HJ 784-2016.
Table 4. Method detection limits for 16 PAHs
| Compound |
MDL (μg/mL) |
HJ 784-2016 MDL (μg/mL) |
| Naphthalene |
0.001 |
0.030 |
| Acenaphthylene |
0.004 |
0.030 |
| Fluorene |
0.015 |
0.050 |
| Acenaphthene |
0.003 |
0.030 |
| Phenanthrene |
0.013 |
0.050 |
| Anthracene |
0.022 |
0.040 |
| Fluoranthene |
0.011 |
0.050 |
| Pyrene |
0.021 |
0.030 |
| Chrysene |
0.010 |
0.030 |
| Benzo[a]anthracene |
0.009 |
0.040 |
| Benzo[b]fluoranthene |
0.008 |
0.050 |
| Benzo[k]fluoranthene |
0.010 |
0.050 |
| Benzo[a]pyrene |
0.012 |
0.050 |
| Dibenzo[a,h]anthracene |
0.007 |
0.050 |
| Benzo[ghi]perylene |
0.009 |
0.050 |
| Indeno[1,2,3-cd]pyrene |
0.007 |
0.040 |
Conclusion
The HPLC method using a Supersil ODS2 column provides reliable separation, excellent linearity, and high sensitivity for 16 priority PAHs in environmental samples. It meets the requirements of HJ 478-2009, HJ 647-2013, and HJ 784-2016, making it suitable for routine monitoring of water, air, and soil.


