Determination of 12 Phenolic Compounds in Environmental Samples by HPLC
- Analyte: 12 Phenols
- System: EClassical 3200 HPLC System with UV Detector
- Column: Supersil Phenyl (5 μm, 4.6 × 250 mm)
- Highlight: Effective separation of 12 phenols using Supersil Phenyl column with baseline resolution of all compounds, meeting HJ 638-2012 requirements.
Introduction
Phenolic compounds are moderately toxic chemicals that can cause chronic poisoning at low concentrations and acute toxicity at high levels. They enter the environment primarily through industrial wastewater from sources such as coking, petroleum refining, chemical synthesis, and plastics manufacturing. Phenols can be absorbed through skin, mucous membranes, and the respiratory tract, posing health risks. Chinese standard HJ 638-2012 specifies an HPLC method for the determination of phenolic compounds in ambient air. This solution presents HPLC method for the analysis of 12 phenols (Phenol, 2-Methylphenol, 3-Methylphenol, 4-Methylphenol, 1,3-Benzenediol, 4-Chlorophenol, 2,6-Dimethylphenol, 1-Naphthol, 2-Naphthol, 2,4,6-Trinitrophenol, 2,4-Dinitrophenol, 2,4-Dichlorophenol) with the Supersil Phenyl column achieving complete separation of all target compounds.
Standards and Reagents
Phenol standards
Phenol, 2-methylphenol, 3-methylphenol, 4-methylphenol, 1,3-benzenediol, 4-chlorophenol, 1-naphthol, 2-naphthol, 2,6-dimethylphenol, 2,4,6-trinitrophenol, 2,4-dinitrophenol, 2,4-dichlorophenol (all ≥99% purity).
Reagents
Methanol (HPLC grade), Deionized water (≥18.2 MΩ·cm),
Other materials
Volumetric flasks, pipettes, membrane filters (0.45 μm), etc.
Standard Solution Preparation
Stock solution (1000 mg/L each): Accurately weigh 0.050 g of each phenol into a 50 mL volumetric flask, dissolve in methanol, and dilute to volume. Store at 4°C in the dark.
Working standard solution (100 mg/L): Pipette 1.0 mL of the stock solution into a 10 mL volumetric flask and dilute to volume with methanol.
Sample Pretreatment
Sample preparation follows HJ 638-2012. For air samples, collect particulate matter on filters and adsorb gaseous phenols on adsorbent tubes. Extract with methanol by sonication or Soxhlet extraction. Concentrate the extract if necessary and filter through a 0.45 μm membrane before analysis.
Instruments and Equipment
HPLC System
EClassical 3200 configured with P3220 high-pressure pump, UV3210 UV-Vis detector, O3220 column oven, Rheodyne 7725 manual injector (or S3210 autosampler optional), Chromatography data station, TP3200 solvent tray
Pretreatment equipment
Solvent filtration apparatus, vacuum pump, ultrasonic cleaner, etc.
Chromatographic Conditions
Mobile phase: A: Acetonitrile; B: Water, in gradient (Table 1)
Table 1. Gradient program
| Time (min) |
A% |
B% |
| 0 |
13 |
87 |
| 5 |
13 |
87 |
| 25 |
35 |
65 |
| 35 |
13 |
87 |
Flow rate: 1.5 mL/min
Detection: UV at 223 nm
Injection volume: 10 μL
Column temp.: 25°C
Experimental Discussion and Results
Separation on Supersil Phenyl Column
Using the Supersil Phenyl column, all 12 phenols were baseline separated (Figure 1). Chromatographic parameters are summarized in Table 2.
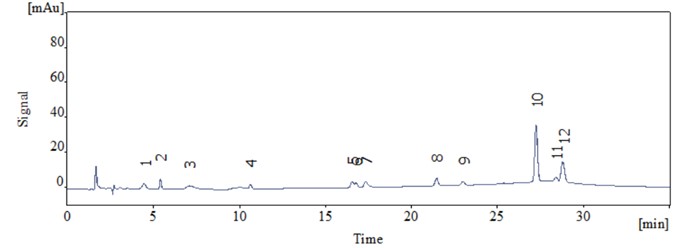
Figure 1. Chromatogram of 12 phenols on Supersil Phenyl column (complete separation)
Table 2. Chromatographic parameters on Supersil Phenyl column
| Peak |
Compound |
RT (min) |
Peak Area (mV·s) |
Asymmetry |
Plate Number (N) |
Resolution |
| 1 |
2,4-Dinitrophenol |
4.46 |
39.80 |
1.01 |
1983 |
– |
| 2 |
1,3-Benzenediol |
5.40 |
56.61 |
1.21 |
15125 |
3.26 |
| 3 |
2,4,6-Trinitrophenol |
7.09 |
25.31 |
1.83 |
11183 |
3.31 |
| 4 |
Phenol |
10.65 |
42.02 |
1.22 |
26142 |
6.42 |
| 5 |
3-Methylphenol |
16.55 |
31.90 |
0.78 |
32567 |
18.78 |
| 6 |
4-Methylphenol |
16.78 |
60.93 |
1.87 |
39636 |
0.65 |
| 7 |
2-Methylphenol |
17.35 |
53.53 |
1.99 |
26695 |
1.52 |
| 8 |
4-Chlorophenol |
21.45 |
26.56 |
1.13 |
73187 |
11.09 |
| 9 |
2,6-Dimethylphenol |
23.00 |
459.82 |
1.02 |
77741 |
4.79 |
| 10 |
2-Naphthol |
27.26 |
135.45 |
1.13 |
131910 |
13.56 |
| 11 |
2,4-Dichlorophenol |
28.40 |
320.60 |
0.25 |
31489 |
2.42 |
| 12 |
1-Naphthol |
28.82 |
39.80 |
1.48 |
81008 |
0.81 |
The Supersil Phenyl column meets the requirements of HJ 638-2012 that offers superior separation, resolving all 12 phenols baseline. The method shows good peak symmetry and high column efficiency.
Conclusion
The HPLC method using either Supersil ODS2 or Supersil Phenyl columns provides effective determination of 12 phenolic compounds in environmental samples. The phenyl column achieves complete separation of all analytes, making it the preferred choice for comprehensive analysis.

