Determination of Prohibited Substances Bromisoval, Carbromal and Carisoprodol in Cosmetics by HPLC
- Analyte: Bromisoval, Carbromal, Carisoprodol
- System: EClassical 3200L HPLC with UV and ELSD Detectors
- Column: Supersil ODS2 (5 μm, 4.6 × 150 mm)
- Highlight: A reliable HPLC-UV/ELSD method for simultaneous determination of three prohibited sedatives in cosmetics
Introduction
With the continuous improvement of cosmetic quality and safety regulations, the detection of prohibited substances has become critical for consumer protection. Bromisoval, carbromal, and carisoprodol are sedatives and muscle relaxants explicitly listed as prohibited ingredients in the Safety and Technical Standards for Cosmetics. Their illegal addition poses potential health risks. According to the national standard GB/T 40899-2021, a high-performance liquid chromatography (HPLC) method has been established for the determination of these three compounds in various cosmetic matrices, including toners, emulsions, and creams. This article presents a complete HPLC solution using the EClassical 3200L system, providing accurate and sensitive quantification for quality control purposes.
Standards and Reagents
Standards: Bromisoval, carbromal, and carisoprodol (purity ≥ 99%)
Other reagents: Acetonitrile, methanol (HPLC grade), deionized water (18.2 MΩ·cm), other solvents and chemicals as required for sample pretreatment (e.g., extraction solvents, filtration membranes)
Standard Solution Preparation
Individual stock solutions: Accurately weigh appropriate amounts of each standard, dissolve in acetonitrile/water (40:60, v/v) or suitable solvent, and dilute to obtain concentrations of approximately 1000 mg/L. Store at 4°C in the dark.
Mixed working solutions: Dilute the stock solutions with the same solvent to prepare a series of calibration standards. For bromisoval and carbromal, concentrations ranging from 10 to 500 mg/L are used; for carisoprodol, the range is 50 to 500 mg/L. All solutions are filtered through a 0.45 μm membrane before injection.
Sample Pretreatment
Sample preparation follows the procedures described in GB/T 40899-2021. Typically, a representative cosmetic sample (e.g. 1–2 g) is accurately weighed and subjected to extraction with an appropriate solvent (e.g., methanol or acetonitrile) under sonication. After centrifugation or filtration, the extract is diluted, cleaned up if necessary, and filtered through a 0.45 μm membrane prior to HPLC analysis. The exact conditions depend on the cosmetic matrix (water-based, emulsion, or cream).
Instruments and Equipment
HPLC System: EClassical 3200L equipped with quaternary pump, UV-vis detector (for bromisoval and carbromal), evaporative light scattering detector (ELSD) (for carisoprodol), autosampler, column oven, solvent bottle tray, Kromstation CDS.
Pretreatment equipment: analytical balance, ultrasonic bath, centrifuge, solvent filtration apparatus, membrane filters (0.45 μm), etc.
Chromatographic Conditions
Column: Supersil ODS2 (5 μm, 4.6 × 150 mm)
Mobile Phase: Acetonitrile / Water = 40 : 60 (v/v)
Flow Rate: 1.0 mL/min
Detection: UV at 210 nm for bromisoval and carbromal:
ELSD for carisoprodol (nebulizer cooling mode, evaporation temperature 40°C, nitrogen carrier gas at 50 psi outlet pressure, flow rate 2.5 mL/min, sensitivity 8)
Injection Vol.: 10 μL
Column Temp.: 30°C
Experimental Results
Standard Separation
Typical chromatograms of standard solutions are shown in Figures 1 and 2. Under the described conditions, bromisoval and carbromal are well resolved with retention times of approximately 2.5 min and 3.2 min, respectively (Figure 1). Carisoprodol elutes at about 4.1 min with good peak shape (Figure 2). No interference from matrix components is observed.
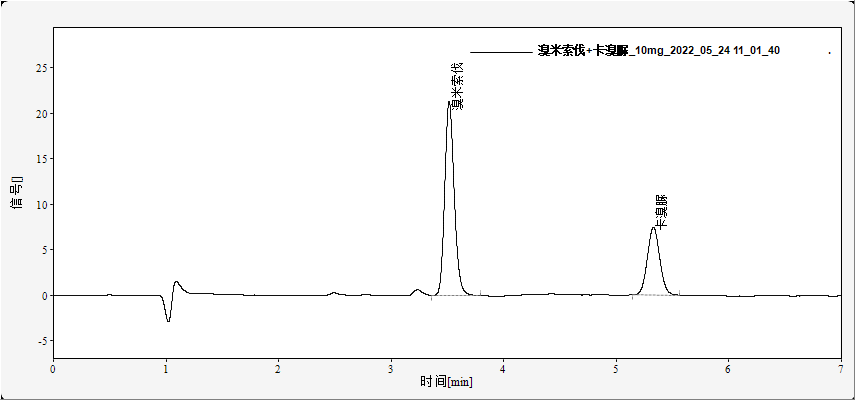
Fig. 1. Chromatogram of bromisoval and carbromal standards (10 mg/L each)
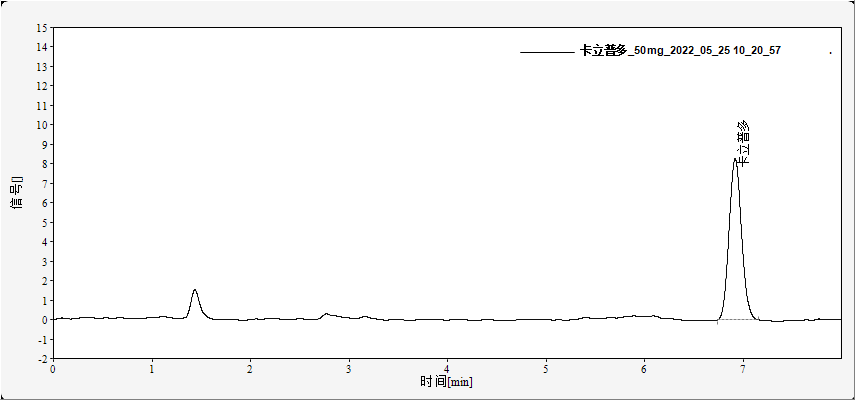
Fig. 2. Chromatogram of carisoprodol standard (50 mg/L)
Method Performance
Linearity and Detection Limit
Linearity: Calibration curves were constructed by plotting peak area versus concentration. For bromisoval and carbromal, excellent linearity was obtained over the range of 10–500 mg/L with correlation coefficients (R²) ≥ 0.9995. For carisoprodol, linearity was established over 50–500 mg/L with R² = 0.9997.
Repeatability: Five consecutive injections of a standard mixture showed relative standard deviations (RSD) for retention time and peak area less than 0.35%, indicating high method precision.
Detection limits (LOD): Based on a signal-to-noise ratio of 3, the method detection limits for the three compounds are listed in Table 1, all well below the requirements of GB/T 40899-2021.
Table 1. Method Detection Limits (S/N = 3)
| Compound |
Method LOD (mg/kg) |
GB/T 40899-2021 Requirement (mg/kg) |
| Bromisoval |
0.27 |
1 |
| Carbromal |
0.78 |
1 |
| Carisoprodol |
3.39 |
10 |
Quantification limits (LOQ): The LOQ (S/N = 10) are 0.9 mg/kg for bromisoval, 2.6 mg/kg for carbromal, and 11.3 mg/kg for carisoprodol (calculated from LOD data, assuming a factor of 3.3; actual values may vary).
Sample Analysis
A hand cream sample was analyzed using the proposed method. Bromisoval was detected at a level of approximately 0.19% (1900 mg/kg), while carbromal and carisoprodol were not detected. This demonstrates the method's applicability for routine screening and quantification of these prohibited substances in complex cosmetic matrices.


