Vitamin A is an essential fat-soluble vitamin that plays critical roles in vision, immune function, and cellular growth. It is commonly found in dairy products, fortified foods, and dietary supplements. Accurate determination of vitamin A is vital for quality control and nutritional labeling. This document presents two HPLC-based analytical methods for vitamin A determination: a reversed-phase method for food samples according to GB 5009.82-2016, and a normal-phase method for vitamin A acetate in pharmaceutical preparations according to the Chinese Pharmacopoeia.
Standards and Reagents
Standard:Vitamin A retinol
Reagents: methanol, ethanol, anhydrous sodium sulfate, potassium hydroxide, ascorbic acid, butylated hydroxytoluene (BHT), petroleum ether, diethyl ether, starch enzyme (amylase) for samples containing starch, deionized water (18.2 MΩ·cm).
Standard Solution Preparation
Vitamin A stock solution (0.500 mg/mL): Accurately weigh 25.0 mg of vitamin A standard, dissolve in anhydrous ethanol, transfer to a 50 mL volumetric flask, and dilute to volume with ethanol. Transfer to an amber glass bottle, seal, and store at –20°C in the dark for up to 1 month. Warm to 20°C before use and correct concentration if needed.
Vitamin A intermediate solution (10.0 μg/mL): Accurately pipette 1.00 mL of vitamin A stock solution into a 50 mL volumetric flask and dilute to volume with methanol. Store at –20°C in the dark for up to 15 days.
Vitamin A series working solutions: Pipette 0.20, 0.50, 1.00, 2.00, 4.00, 6.00 mL of intermediate solution into separate 10 mL amber volumetric flasks and dilute to volume with methanol to obtain concentrations of 0.20, 0.50, 1.00, 2.00, 4.00, 6.00 μg/mL. Prepare fresh before use.
Sample Pretreatment
Sample preparation:Homogenize the sample and store in amber bottles at cool temperature, protected from light. Analyze as soon as possible.
For samples without starch: Weigh 2–5 g (solid, to 0.01 g) or 50 g (liquid, to 0.01 g) into a 150 mL flask. For solid samples, add about 20 mL warm water and mix. Add 1.0 g ascorbic acid and 0.1 g BHT, mix. Add 30 mL anhydrous ethanol and 10–20 mL potassium hydroxide solution, shaking while adding. Saponify in an 80°C water bath with shaking for 30 min. Cool immediately in cold water.
For samples containing starch: Weigh sample as above, add about 20 mL warm water, add 0.5–1 g amylase, incubate in a 60°C water bath with shaking for 30 min. Then add ascorbic acid, BHT, ethanol, and KOH as above and saponify.
- Extraction: Transfer the saponified solution to a 250 mL separatory funnel with 30 mL water. Add 50 mL petroleum ether-diethyl ether mixture, shake for 5 min. Transfer the lower layer to another separatory funnel and repeat extraction with 50 mL of the ether mixture. Combine the ether layers.
- Washing: Wash the combined ether layer with about 100 mL water, repeating 3 times until neutral (check pH of lower layer with pH paper). Discard the aqueous phase.
- Concentration: Pass the ether layer through anhydrous sodium sulfate (about 3 g) into a 250 mL evaporation flask. Rinse the funnel and sodium sulfate with 15 mL petroleum ether twice. Evaporate at 40°C under vacuum or nitrogen flow to about 2 mL, then blow to near dryness with nitrogen. Dissolve the residue in methanol, transfer to a 10 mL volumetric flask, and dilute to volume with methanol. Filter through a 0.45 μm organic membrane before HPLC analysis.
Instruments and Equipment
HPLC System: EClassical 3200 equipped with P3200 high-pressure pump, D3210 UV-Vis detector (or fluorescence detector), S3210 autosampler, O3220 column oven, T3200 solvent bottle tray, Kromstation CDS.
Pretreatment equipment: Analytical balance, centrifuge, ultrasonic cleaner, vortex mixer, nitrogen evaporator, water bath, pH meter, separatory funnels, volumetric flasks, etc.
Chromatographic Conditions
Column: Supersil ODS2 (5 μm, 4.6 × 250 mm)
Mobile Phase: A-water; B-methanol (in gradient)
Flow Rate: 0.8 mL/min
Detection: UV at 325 nm, or fluorescence with excitation at 328 nm and emission at 440 nm
Injection Volume: 10 μL
Column Temp.: Ambient
Standard Separation
A typical chromatogram of a vitamin A standard (retinol) analyzed under the reversed-phase conditions is shown in Figure1 of the original document. The peak is well-resolved and suitable for quantitative analysis.
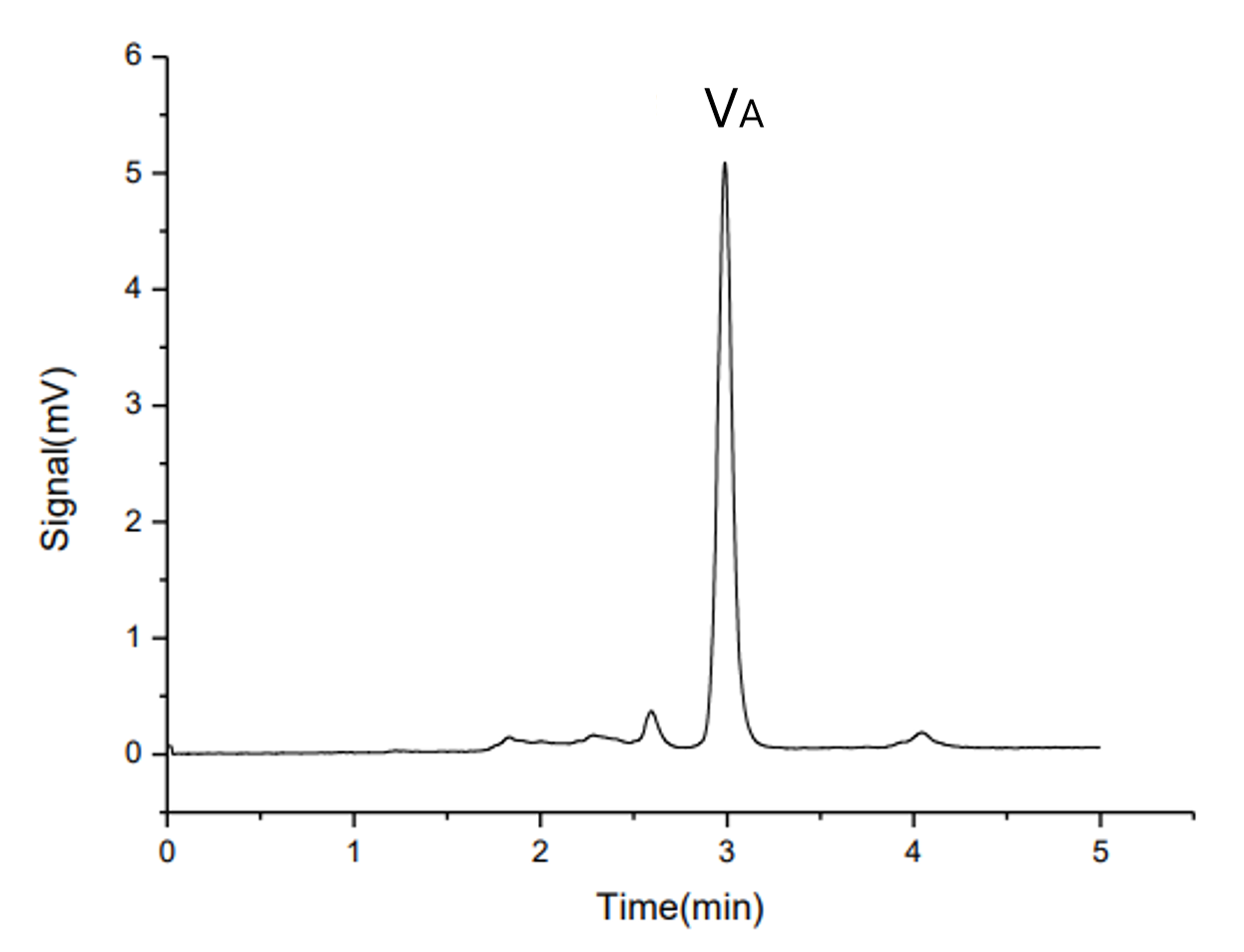
Fig. 1. Chromatogram of Vitamin A standard solution (at UV325nm)
Standards and Reagents
Vitamin A standard: Vitamin A acetate reference standard
Reagents: n-Hexane (HPLC grade)
Standard Solution Preparation
Vitamin A acetate reference solution: Accurately weigh about 45 mg of vitamin A acetate reference standard into a 100 mL amber volumetric flask, dilute to volume with n-hexane, and mix. Pipette 5 mL of this solution into a 50 mL amber volumetric flask and dilute to volume with n-hexane.
Sample solution: Accurately weigh about 0.5 g of sample into a 50 mL amber volumetric flask and dilute to volume with n-hexane.
Sample Pretreatment
For pharmaceutical-grade materials, no further pretreatment is typically required. Prepare the sample solution as described above.
Instruments and Equipment
HPLC System: EClassical 3200 (same configuration as above)
Pretreatment equipment: Analytical balance, volumetric flasks, etc.
Chromatographic Conditions
Column: Silica gel column (5 μm, 4.6 × 250 mm)
Mobile Phase: n-hexane/isopropanol, 998/2 (v/v)
Flow Rate: 0.8 mL/min
Detection: UV at 325 nm
Injection Volumn:10 μL
Column Temp.:Ambient
Standard Separation
A typical chromatogram of a vitamin A acetate standard analyzed under normal-phase conditions is shown in Figure2. The method separates vitamin A acetate from its cis-isomer. Chromatographic parameters are summarized in Table 1.
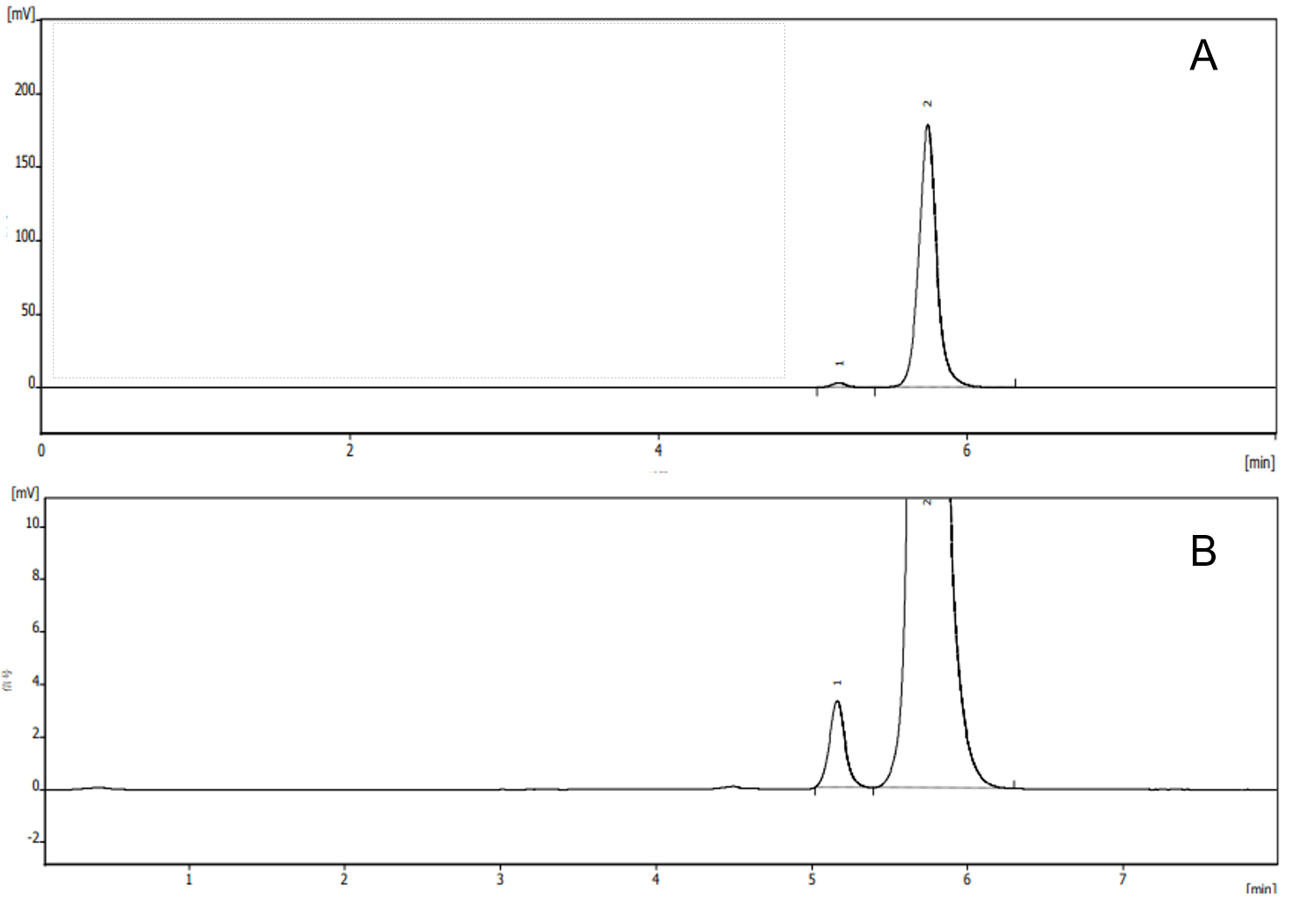
Fig. 2. Chromatogram (A)and enlarged chromatogram (B) of Vitamin A acetate standard solution (at UV325nm)
Table 1. Chromatographic Parameters for Vitamin A Acetate (Normal-Phase)


